Observational data have demonstrated that percutaneous transcatheter closure of patent foramen ovale reduces the risk of cryptogenic and cardiac stroke.1
The patient can go back home on the same day or early next morning after PFO closure however the recovery takes 2-6 weeks to completely resume a normal routine.2
PFO closure has found to have an increased risk on new onset of atrial fibrillation however it occurs early and is transient.3
How Safe Is PFO Closure What Is The Recovery Time For It?
Although patent foramen ovale is a medical procedure yet it is neither invasive nor leaves a scar after the procedure. However, it is important to remember that there are risks involved. Some of the complications include ischemic stroke as a result of the procedure, atrial fibrillation, and bleeding in the groin region where the device was inserted.
In rare instances, it may cause blood clots in the legs or lungs and leave an injury in the heart. The catheterization procedure is usually performed at a clinic or hospital by professionals and you will be kept in observation overnight. If the doctor sees no complications and you have completely worn out of the sedation, you will be suggested to go home. Several studies have been performed to determine the complications of the two devices used for PFO closure. Most clinical trials have shown that PFO closure has no risk associated with it and is considered to be safe.1
However, the patient can see a tenderness or bump at the procedure site. You may experience bounced heartbeat, discomfort, fatigue, and pressure levels fluctuating for the next 2-6 weeks. These are common symptoms and don’t require treatment. But there can be activity restrictions such as
- No heavy lifting
- No heavy activities for the next 6-8 weeks
- Avoid driving for the first one week
- Only shower and no tub baths or swimming
Most patients recover in a week and resume work. However, if you experience severe discomfort, bleeding, or infection report immediately.2
Can PFO Closure Cause AFIB?
A new meta-analysis of patients undergoing PFO closure after a cryptogenic stroke or migraine is linked with an enhanced risk of atrial fibrillation. Studies in the past have found an increased risk of the onset of atrial fibrillation as high as 3% after the procedure.
More than three-fourth of device-associated AFIB have been resolved without multiple recurrences. In a nutshell, the events generally appear early and resolve without recurrence.
A study was conducted on 3,924 patients with either cryptogenic stroke or migraine, the overall prevalence of atrial fibrillation in 8 randomized trials demonstrated no recurrence in 76% of this population.
Most of the cases got resolved either spontaneously or through simple therapies. Always paroxysmal, AF occurred only in the first-month post-PFO closure, and AECG at 6 months follow-up after PFO closure, was relatively low (<5%) with no adverse outcomes. The primary objective of this study is to define an irregular rhythm during the initial six months.3
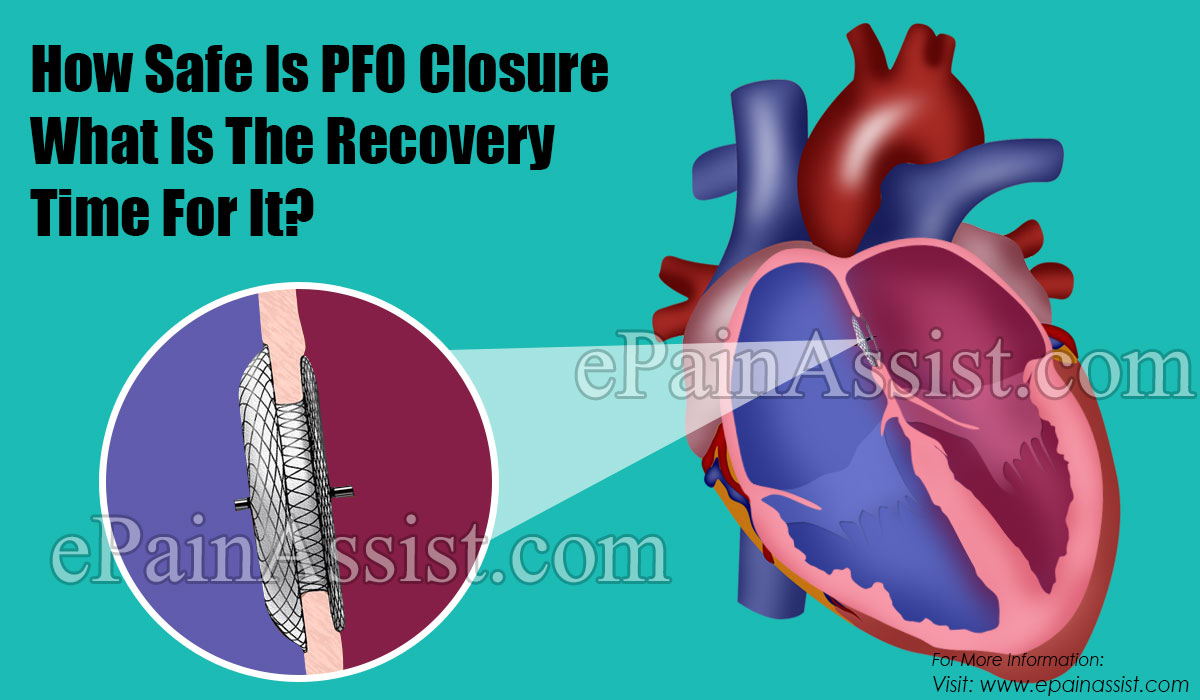
PFO was initially considered as a manifestation of ASD however it exhibited properties in adults and also represented the easiest target for closure. The most widely used procedure for PFO closure is cardiac catheterization wherein a thin device is inserted for the closure.
But CardioSEAL is the most used device in the United States as this has the best implantation technique.
- Luermans, J G L M, et al. “Complications and Mid-Term Outcome after Percutaneous Patent Foramen Ovale Closure in Patients with Cryptogenic Stroke.” Netherlands Heart Journal : Monthly Journal of the Netherlands Society of Cardiology and the Netherlands Heart Foundation, Bohn Stafleu Van Loghum, Oct. 2008, www.ncbi.nlm.nih.gov/pmc/articles/PMC2570764/.
- “Patent Foramen Ovale Transcatheter Repair.” Johns Hopkins Medicine, www.hopkinsmedicine.org/health/treatment-tests-and-therapies/patent-foramen-ovale-transcatheter-repair.
- “Atrial Fibrillation After PFO Closure – Full Text View.” Atrial Fibrillation After PFO Closure – Full Text View – ClinicalTrials.gov, clinicaltrials.gov/ct2/show/NCT04290052.
Also Read:
- What is Patent Foramen Ovale (PFO): Causes, Symptoms, Treatment, Diagnosis
- Can I Exercise With A Patent Foramen Ovale & Are There Any Home Remedies?
- How Do You Fix A Patent Foramen Ovale & Can PFO Cause A Heart Attack?
- What Happens If The Patent Foramen Ovale Fails To Close & Can You Live With A PFO?
- Does Patent Foramen Ovale Run In Families & Does A PFO Cause Fatigue?
